#ScienceSaturday posts share exciting scientific developments and educational resources with the KAND community. Each week, Dr. Dylan Verden of KIF1A.ORG summarizes newly published KIF1A-related research and highlights progress in rare disease research and therapeutic development.
KIF1A-Related Research
RAB3 phosphorylation by pathogenic LRRK2 impairs trafficking of synaptic vesicle precursors
During the COVID-19 epidemic many of us experienced how devastating shipping disruptions were; industries with diverse products all encountered the same bottleneck, and customers with diverse needs were left without. Like shipping disruptions, the disruption of axonal cargo trafficking can have many causes and many effects, depending on the primary cause.
While we usually think of this cause as KIF1A mutations, upstream regulators of motor proteins can also disrupt traffic. In a recent pre-print*, the Holzbaur Lab at University of Pennsylvania investigated how a protein called LRRK2 regulates the traffic of synaptic vesicle precursors, a type of KIF1A cargo.
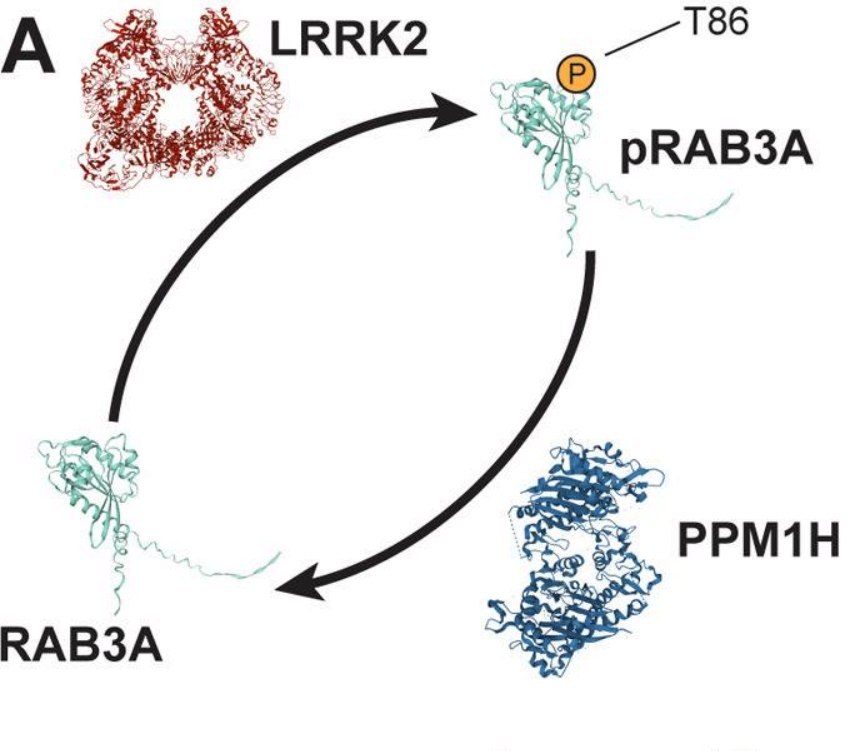
LRRK2, PPM1H, and RAB3A
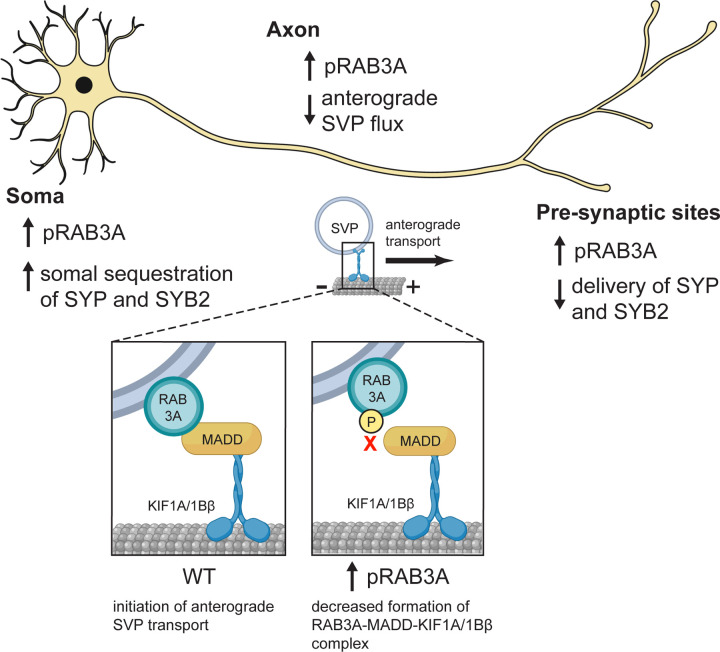
LRRK2 is actually related to another neurodegenerative motor disorder, as the most common causes of genetic Parkinson’s Disease. Recent work by the Holzbaur lab found that gain-of-function mutations that over-activate LRRK2 disrupt axonal transport toward the cell body. In the present study, they found that overactive LRRK2 also disrupts the transport of synaptic vesicle precursors away from the cell body, mediated by kinesins. Notably these cargo weren’t moving slower or less reliably, there were just less entering the axon; this indicates an issue with connecting cargo to the motor protein, rather than affecting the motor’s ability to move.
How does LRRK2 regulate axonal traffic? Well it’s part of a team:
- RAB3A is a protein whose behavior depends on whether or not it has a phosphate attached to it (we’ll call this pRAB3A). As we’ll discuss, RAB3A interacts with other proteins that regulate axonal cargo transport
- LRRK2 adds a phosphate group to RAB3A
- PPM1H removes a phosphate group to RAB3A
The Study
By genetically over-activating LRRK2, or knocking out PPM1H, the authors were able to increase the amount of pRAB3A and disrupt synaptic vesicle precursor transport. They could improve transport by temporarily preventing the attachment of phosphate to RAB3A.
RAB3A is known to interact with another protein called MADD, which regulates the attachment of synaptic vesicle precursor cargo to KIF1A and KIF1B. But pRAB3A is unable to bind to MADD, which is a bit like not having a hitch between the semi truck and its container.
It’s important to remember that proteins like LRRK2 regulate many pathways, including those unrelated to transport by kinesins, and KIF1A carries cargo other than synaptic precursor vesicles. But looking at other disorders that impact specific facets of KIF1A function can teach us a lot about KIF1A and KAND, especially as LRRK2 inhibitors are investigated for Parkinson’s Disease.
*What’s a pre-print? Check out this #ScienceSaturday post to learn more.
Want to learn more about KIF1A work at the Holzbaur Lab? Check out Dr. Jayne Aiken’s talk from our 2023 Scientific & Family Engagement Conference.
Rare Roundup
Patient advocacy groups from around the world convene at the CZI Science in Society Conference and the Global Genes Rare Advocacy Summit
This week the rare disease community was blessed with an overabundance of expertise, storytelling, empathy, and learning thanks to two organizations that aim to consolidate and empower the efforts of rare disease groups. KIF1A.ORG team members attended both virtually and in-person, making new connections that we’re excited to explore and share with you.
Here are a few of the themes discussed over the course of this week:
- The interface between groups representing rare genetic disorders, and broader diseases with overlapping cases
- Using collective data to accelerate diagnosis and find therapeutic opportunities
- Creating equitable access to rare disease information and clinical opportunities
- Using technology to make research and clinical engagement less burdensome for patients
The coordination it takes to foster productive conversations between so many stakeholders can’t be overstated, and while we’ve rounded up both conferences in this segment, we want to express our gratitude to both CZI and Global Genes teams for gathering us to grow together.

