#ScienceSaturday posts share relevant and exciting scientific news with the KAND community. This project is a collaboration between KIF1A.ORG’s Research Engagement Team Leader Alejandro Doval, President Kathryn Atchley, Science Communication Volunteer Aileen Lam and Chief Science Officer Dr. Dominique Lessard. Send news suggestions to our team at impact@kif1a.org.
KIF1A-Related Research
Planar cell polarity protein Vangl2 and its interacting protein Ap2m1 regulate dendritic branching in cortical neurons
If you’ve ever seen a picture of a neuron, you might have notices that this cell type is… kind of a funny shape? On one end of a neuron you have a round cell body with many dendrites branching off that look like tree branches. On the other end you have a long skinny protrusion called the axon that leads to the synaptic terminals, a part of the neuron that helps send messages to other cells. Why does one end of a neuron look so much different than the other end? The answer lies in the concept of cell polarity. Cell polarity is defined as the “asymmetric organization of several cellular components.” In many cell types, this asymmetry is important for specific cellular functions and in the case of neurons, it’s very important for transmitting signals within and between cells.
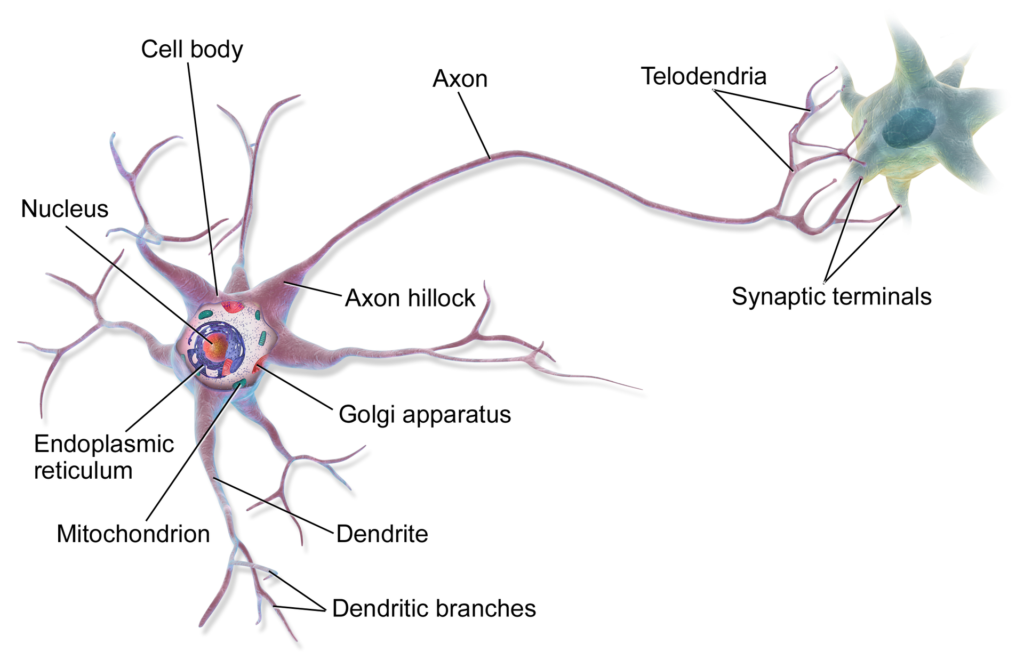
What causes cell polarity in neurons? It turns out that a lot of proteins in the neuron play a role in this process. The paper we are featuring today focuses on the role of a protein called Van Gogh-like 2 (Vangl2). To further understand how Vangl2 plays a part in determining cell polarity, the authors screened (looked for) additional proteins that are known to interact with Vangl2. As you might guess, one of the proteins that was found to interact with Vangl2 was KIF1A! Identifying KIF1A and other proteins that interact with Vangl2 helped the authors better understand neuronal cell polarity, specifically the development of dendrites. Advancing our understand of neuronal systems by identifying the many roles that KIF1A plays within a neuron are critical to advancing our understanding of the impact of KIF1A variants in human disease. Want to learn more about cell polarity? Check out the video below!
Rare Roundup
The positive impact of sharing negative research results
When reading summaries of scientific findings, many articles and publications will only share the positive, or hypothesis (idea) confirming, results to tell a scientific story. If we are looking for answers to complex problems in rare disease, shouldn’t we be focused on the parts of studies that show significant change in experiments? Not necessarily. The article we are sharing today discusses the importance and the impact of scientific authors sharing negative results, or results that may prove an original hypothesis to be incorrect. The article goes on to discuss how we can learn just as much, if not more, from what didn’t work than what did work. This is especially important in the field of rare disease research where the absence of information, be it positive or negative, can have deleterious effects on therapeutic development. Want to learn more about the importance of negative results in the rare disease space? Click the button below to read the article!
“…The need to share negative information is gaining backers, but in a rare disease field such as neuromuscular disorder research, not having all the information is a tragedy.”
Dr. Arechavala-Gomeza
Lab-grown ‘mini brains’ hint at treatments for neurodegenerative diseases
This article summarizes how researchers at Cambridge have created brain organoids, or “mini brains,” to model neurodegenerative diseases ALS and frontotemporal dementia (FTD). Modeling diseases isn’t new. Our KIF1A Research Network uses a variety of model systems—like mice, induced pluripotent stem cells (iPSCs), zebrafish and even worms—to better understand KIF1A/KAND. This article caught our attention because it discusses the complexity that scientists have to navigate when working with model systems.
- The pros and cons of working with animal models (a full living organism, but not human) vs. cell-based models (not a full living organism, but human)
- Working with a cell line, mouse, etc. that is 1 day old or 1 week old is very different than working with models at 1 or 2 years old (but longer lived models obviously take more time to generate and are much, much, much more expensive to study)
- If you think keeping houseplants alive is a challenge, it’s even more difficult to keep model systems alive and well—even iPSCs and organoids need continuous, precisely managed nutrition to grow and survive
Check out the article to learn more about this research and common challenges scientists have to work through when modeling diseases.
NIH study suggests people with rare diseases face significantly higher health care costs
In other breaking news, water is wet… A study led by the National Institutes of Health’s National Center for Advancing Translational Sciences (NCATS) was just published with new evidence to vouch what patients and caregivers in the rare disease community already know: living rare is expensive.
“In every case, the cost per patient per year (PPPY) for those with a rare disease exceeded costs for non-rare diseases patients of the same age. According to the Eversana healthcare system database … PPPY costs ranged from $8,812 to $140,044 for rare diseases patients compared to $5,862 for those without a rare disease. The NCATS data, which drew from estimates mostly from Florida Medicaid information over five years, indicated PPPY costs ranging from $4,859 to $18,994 for rare diseases patients versus $2,211 for those without a rare disease.”
While these findings won’t come as a surprise to the rare disease community, it is much needed to convince decision-makers in government and the healthcare, insurance and pharmaceutical industries to take action to address the significant unmet needs of rare disease patients.

