#ScienceSaturday posts share relevant and exciting scientific news with the KAND community, and are compiled by Alejandro Doval. Alejandro is from Spain and serves as Team Leader of the KIF1A.ORG parent-led Research Engagement Team. Send news suggestions to our team at impact@kif1a.org.
Recent KIF1A-Related Research
Smart motors and cargo steering drive kinesin-mediated selective transport
An abstract of kinesin research conducted at Rensselaer Polytechnic Institute in New York was recently published in Molecular and Cellular Neuroscience.
“Two main models have been proposed to explain kinesin selectivity. In the smart motor model, kinesin selectivity is conferred by a preference of the kinesin motor domain for specific subsets of microtubules. In the cargo steering model, kinesin selectivity is modulated by the vesicular cargo to which the motor is bound. We evaluate the evidence for both models and conclude that while the smart motor model may explain axonal selectivity, cargo steering is required for dendritic selectivity.”
We’ll report back when we learn more about this paper.
Bonus: What Is Kinesin?
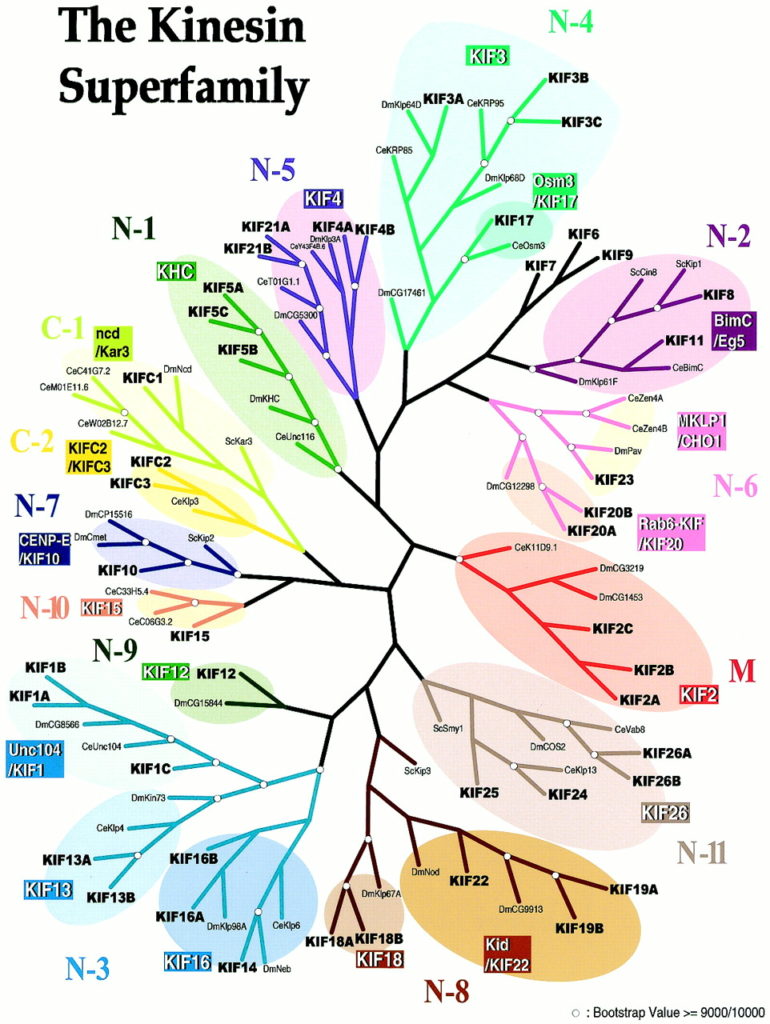
We know that KIF1A belongs to the kinesin superfamily of genes (KIFs)—a family of 46 members!
KIFs are motor proteins that transport cargo along the highway of cells, microtubules. KIF researcher Dr. Ronald Vale explains why KIFs are so important to healthy cell function in this helpful video.
Rare Disease News
First up, we have two encouraging updates in therapeutic development for rare diseases.
Risdiplam (RG7916) Pivotal FIREFISH Study Demonstrated Statistically Significant Improvement in Infants with Type 1 Spinal Muscular Atrophy
“The pivotal study assessed the efficacy of risdiplam (RG7916) in infants with type 1 spinal muscular atrophy (SMA), the most severe, infantile onset form of this rare and devastating neuromuscular disease. Risdiplam has been well tolerated and no treatment-related safety findings leading to withdrawal have been observed in any risdiplam trial to date. Data from part 2 of the FIREFISH study will be shared with health authorities globally and will be presented at an upcoming medical congress.
‘We are excited about the FIREFISH results as they demonstrate how effective risdiplam is in type 1 SMA patients, where developmental milestones such as rolling, sitting and standing were achieved,’ said Stuart W. Peltz, Ph.D., Chief Executive Officer of PTC Therapeutics. ‘These results further support the growing body of evidence of risdiplam’s benefit in SMA patients across all types studied and reinforce the potential of our small molecule splicing platform to identify new therapies for patients who have limited or no treatment options.’”
Alberta baby ‘Mighty Hudson’ home after gene therapy: ‘he’s cured’
“Hudson was diagnosed with Severe Combined Immunodeficiency (SCID) within days of his birth on June 23. The condition, known to many as ‘Bubble Boy Disease,’ prevents his body from fighting illnesses. Essentially, he didn’t have an immune system.
In August, Hudson was accepted into a gene therapy program at St. Jude Children’s Research Hospital in Memphis. A medical team removed some of his bone marrow cells and replaced the faulty gene with a corrected one. The cells were placed back into his body through an IV.”
“Medicine is amazing. We took a chance on science and we’re so glad that we did. It’s amazing what they’re able to do.”
–Hudson’s dad
Researchers uncover mechanism for how common gene therapy vectors enter cells
“Researchers led by a team at Massachusetts Eye and Ear have identified a novel cellular entry factor for adeno-associated virus vector (AAV) types—the most commonly used viral vectors for in vivo gene therapy.”
“This finding may enable scientists to better direct AAV gene transfers to targeted cell tissues, in order to treat specific genetic diseases.”
– Luk Vandenberghe, Ph.D., Senior study author
This story provides a helpful description of how AAV vectors can be used in gene therapy, and how these researchers are working to improve the safety and effectiveness of AAVs.