Authored by KIF1A.ORG Volunteer Roberto Ogelman, PhD
Heterogeneity in research
When we talk about KIF1A-associated neurological disorder (KAND), it can be tempting to think of as a single condition with a single cause. But in reality, KAND is caused by a spectrum of changes, or mutations, to the KIF1A gene.
These different mutations can lead to different outcomes depending on how they affect the function of the KIF1A protein.
In science, this is called heterogeneity, meaning that different genetic changes in the KIF1A gene can lead to different biological effects, and ultimately, different experiences for individuals and families.
The importance of equity in a diverse disorder
Because KAND is heterogeneous, there is a risk that some groups of patients are studied more than others. This means that certain populations may not be equally reflected in research studies and therapeutic development.
An equitable approach means a wide range of mutations are studied, not just the more common ones. This ensures diverse patient representation and helps researchers develop treatments that consider the full spectrum of KIF1A mutations.
A recent pre-print by Dr. Jayne Aiken, Assistant Professor at Colorado State University, and colleagues helps us better understand why this diversity matters, and why studying different mutations is essential for moving towards better treatments.
A word from Jayne Aiken, PhD
Our study focuses on KIF1A variants selected to reflect a range of motor changes seen across the KAND community, and how these changes impact neuron development and the transport of important materials to synapses. Understanding these differences helps explain each patient’s experience and supports more personalized, patient-centered research and care.
What did this study measure, and why does it matter?
Neurons rely on small structures called synapses to help send and receive information. These connections are crucial for our ability to learn, form memories, and process the world. And synapses depend on cargo transported by KIF1A.
To understand heterogeneity in KAND, researchers measured how different KIF1A mutations affect the formation and function of these synapses.
Focusing on how different changes to the KIF1A gene may have a range of effects on neurons is an important step towards reaching more equitable KAND research.
Note: While this study advances our knowledge of how different KIF1A mutations impact neuron development, there are many KIF1A mutations that are not represented. Even mutations that fit in the same broad category of mutation may have drastically different effects on neurons and synapses. Future studies will expand on this representation.
Not all KIF1A mutations are the same
One of the most important insights from Dr. Aiken and colleagues is that different KIF1A mutations affect cells in fundamentally different ways.
Researchers grouped the KIF1A mutations they studied into three broad classes:
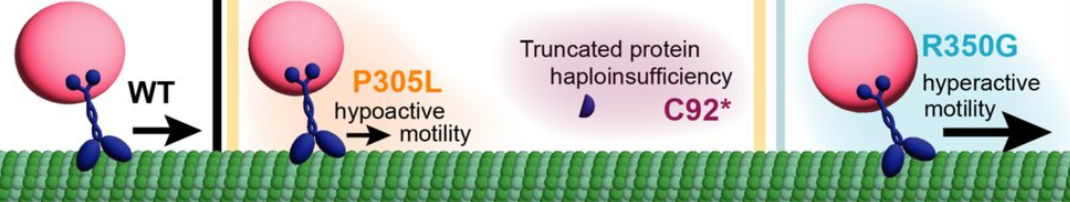
1. Null/Haploinsufficient mutations (less working protein)
These mutations, like C92*, prevent one copy of the KIF1A protein from being made or functioning at all; the remaining copy has to pick up the workload, like a shortage of drivers.
- Effect on neurons and synapses: While neurons survive normally, the C92* mutation results in a reduction in neuron outgrowth and an overall decrease in synaptic communication.
- Therapeutic context: Symptoms may be milder or show up later in life but can still cause progressive damage to neurons over time. For patients with null mutations, our Research Network is studying how to increase levels of healthy KIF1A.
2. Hypoactive mutations (too slow or inefficient at transporting cargo)
Mutations like P305L produce a motor that still moves along microtubules, but not as well. KIF1A doesn’t get cargo to where it needs to go, and may block traffic for other motors as well.
- Effect on neurons and synapses: The P305L mutation increases cell death during development of neurons. Neurons that do survive show a decrease in overall outgrowth and synaptic communication.
- Therapeutic Context: Hypoactive mutations are common in KAND. Often a single mutation interferes with the healthy copy of KIF1A; patients may benefit from therapies that reduce expression of the hypoactive KIF1A, like the ASO therapy developed by n-lorem.
3. Hyperactive mutations (too fast or overactive)
Mutations like R350G make KIF1A move too fast or too much. While it might not sound bad to have KIF1A moving too well, these motors can overshoot their targets, like a bus skipping stops on its route.
- Effect on neurons and synapses: While neuron survival and outgrowth were normal, KIF1A was less likely to deliver cargo to specific locations on the axon.
- Therapeutic Context: For mutations like these it may take two copies to cause disease, so reducing mutant expression may not be viable. Directly editing the KIF1A gene to correct these mutations could be the most viable direct approach.
Why heterogeneity matters for families and research
Understanding diversity in KIF1A mutations is important to study because it allows for more comprehensive and personalized approaches to treat KAND.
1. Better Diagnoses – Recognizing that different mutations behave differently helps clinicians interpret genetic results and anticipate possible outcomes. This can support earlier interventions and more informed care decisions.
2. More targeted treatments – A therapy that helps one type of mutation may not help another. Knowing how different changes to KIF1A behave allows for specific treatment approaches to be used case-by-case, improving outcomes for patients.
Most importantly, understanding this heterogeneity bring us closer to meeting the needs of every individual and family in the KIF1A community.
Summary by Roberto Ogelman, PhD
Dr. Ogelman completed his PhD in Neuroscience at the University of Colorado – Anschutz Medical Campus in 2024, with a focus on neuronal development. He is excited to be joining the Dravet Syndrome Foundation in May 2026 as their Scientific Programs Manager.

