KIF1A.ORG’s 16th Research Roundtable meeting, “Disease-associated heterodimers are more toxic than homodimers” was presented by Dr. Shinsuke Niwa at Tohoku University in Japan.
Attendance

23 RESEARCHERS, CLINICIANS, & BIOTECH REPS

19 RESEARCH INSTITUTIONS & INDUSTRY PARTNERS/ORGS
5 KIF1A.ORG REPS
Who is Dr. Niwa?

Dr. Niwa is an assistant professor at the Frontier Research Institute for Interdisciplinary Sciences at Tohoku University in Sendai, Japan. His career has included extensive research into kinesin function, using cell and worm models to bridge basic research and therapeutic relevance. His studies address many common questions regarding KAND mechanisms, including how different mutations cause slower or faster motor function, and how mutations in a single copy of the KIF1A gene impact protein function. You can get to know more about Dr. Niwa here.
Summary
“Disease-associated heterodimers are more toxic than homodimers”
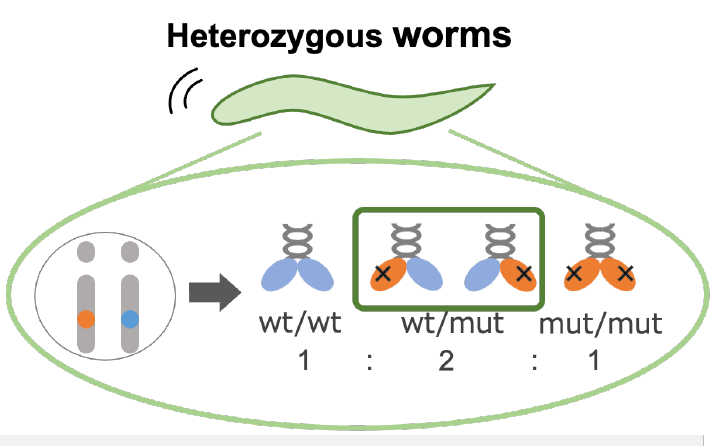
- KIF1A is a motor protein that operates in pairs, called dimers, in order to move cargo up and down neuronal axons. Because many KAND patients carry only one mutant copy of the KIF1A gene, three kinds of KIF1A dimers coexist: Pairs of healthy KIF1A, pairs of mutant KIF1A, and pairs with both healthy and mutant KIF1A called heterodimers. While we know that mutations in KIF1A can reduce function, the ability of heterodimers to transport cargo in neurons is relatively unknown.
- Dr. Niwa’s group studied KIF1A function in worms called C. elegans, which are easy to mutate and have transparent bodies, allowing analysis of KIF1A mutants in live animals. They generated mutations in worms so that they carried one gene copy of healthy kif1a, and one gene copy of mutant kif1a that was based on mutations seen in KAND.
- Dr. Niwa found that heterodimers formed from healthy and mutant KIF1A have a reduced ability to travel along axons, resulting in mislocalization of cargo. Imagine walking down the road with your left leg in a cast – even though your right leg is healthy, you need them both to move quickly and effectively to your destination.
- C. elegans is a powerful model in part because random mutations can be introduced. Dr. Niwa’s group induced secondary mutations in several C. elegans lines already carrying different KAND-associated mutations, selecting worms whose movement improved. While we don’t recommend creating additional mutations in humans, we can learn a lot about how disease-associated mutations damage KIF1A function by observing which other mutations impact the organism.
Main Takeaways
- While worms are not exact models of human disorders like KAND, they are a valuable tool to explore scientific questions that we can’t experiment with on humans or would take more time and money to explore in other model systems like mice.
- “Basic” animal models like worms can provide huge insight into how we should approach therapeutic development for KAND. Dr. Niwa’s work sparked a rich discussion about how we might pursue further research based on his new findings.
- Healthy and mutant KIF1A proteins associate with one another. Decreasing the ratio of mutant:healthy KIF1A may help cargo trafficking.
- Read more of Dr. Niwa’s research here.

